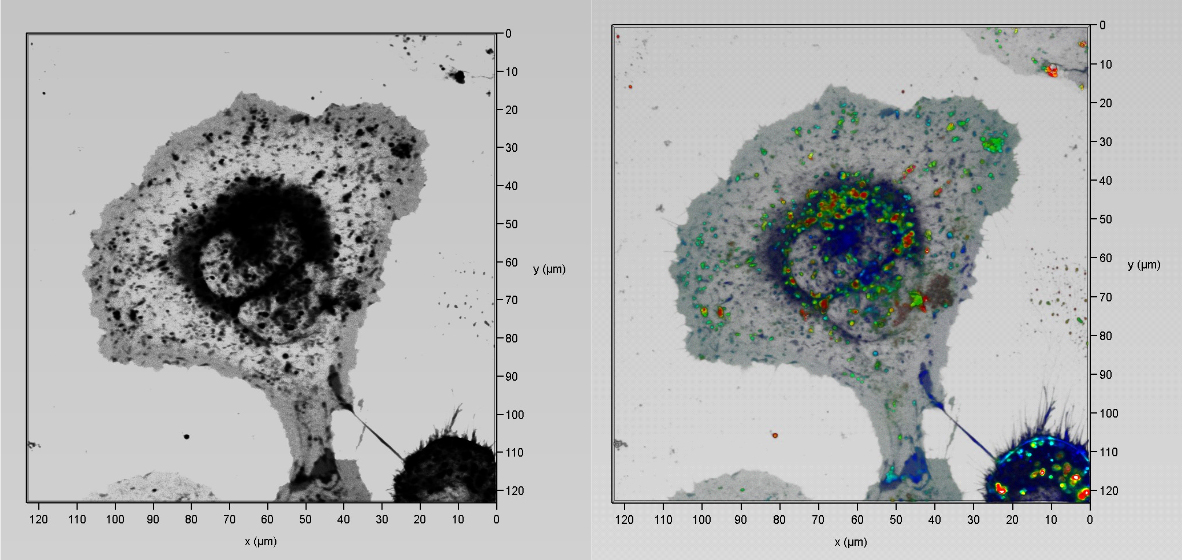
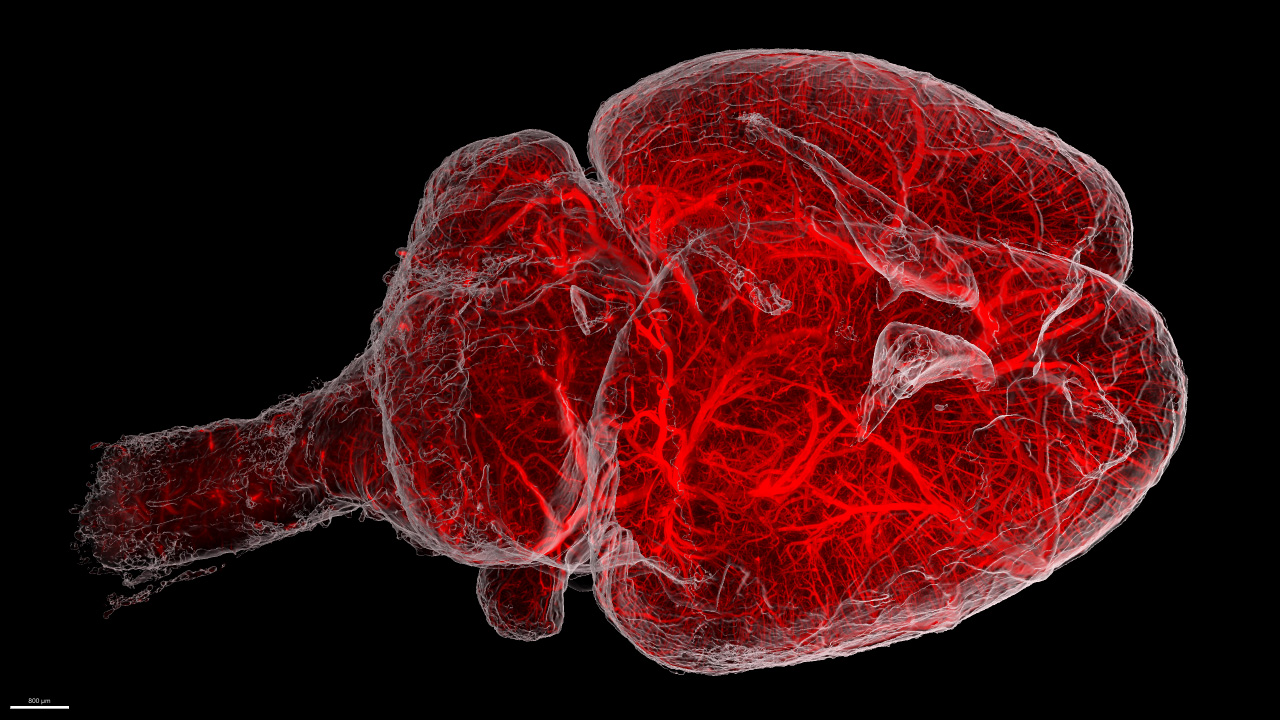
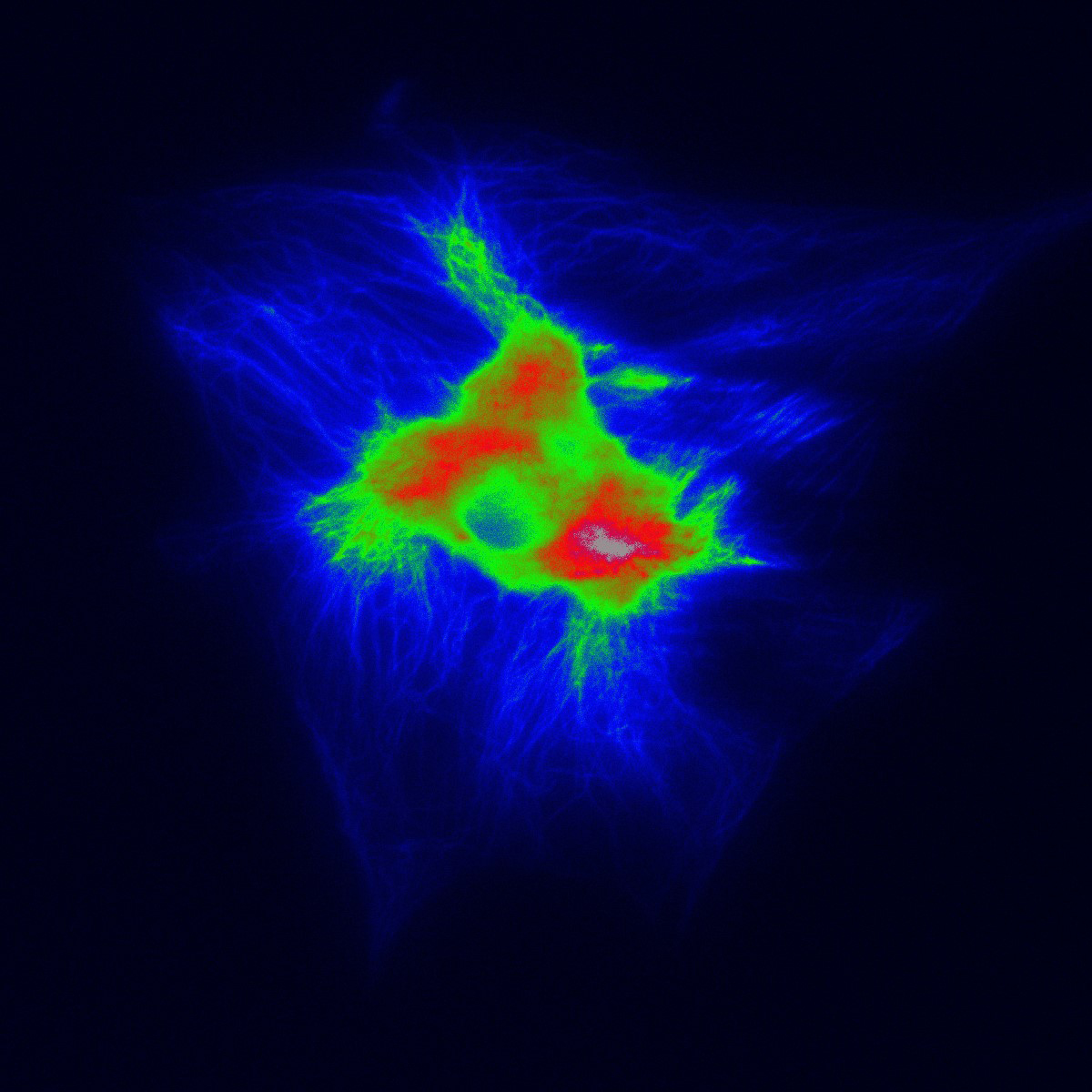
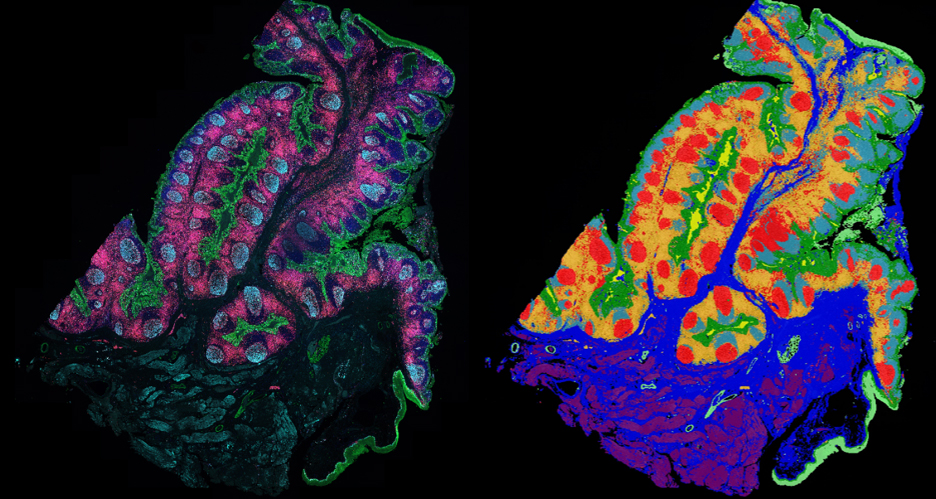
Clearing tissue is now a powerful and accessible technique for studying thicker samples in greater detail. The recent proliferation in tissue-clearing reagents and methods has made the technique more effective and more reliable over a greater range of samples from tissues to organs to whole animals. Once a suitable tissue-clearing regime has been established for a sample the following consideration is what image acquisition modality to use? Two potential options are a spinning disk confocal and a lightsheet microscope, both of which we will present in this workshop. Some of the parameters of a sample or the feature of interest will clearly direct this decision; for example, imaging a whole cleared mouse would typically preclude a spinning disk confocal and imaging a cleared tissue sample at super-resolution would typically preclude lightsheet microscopes. When imaging cleared specimens multiple factors need to be taken into consideration, foremost is a careful evaluation of the volumetric data such that it meets resolution requirements and is an accurate 3D representation of the sample. Other considerations are practical including: safety and ease of sample handling, ease of capture and duration of data acquisition. In this workshop we present our Marianas SDC inverted microscope system featuring a CSU-W SoRa super-resolution spinning disk confocal and our Cleared Tissue LightSheet (CTLS) microscope system and explore cleared tissue samples that could be imaged by each modality and compare the benefits and limitations of each technique.
Workshop 6
THU 9 JUNE 2022, time: 16:30 - 17:30
Click on the images to open the full image view!
Abbelight
Stand 36b
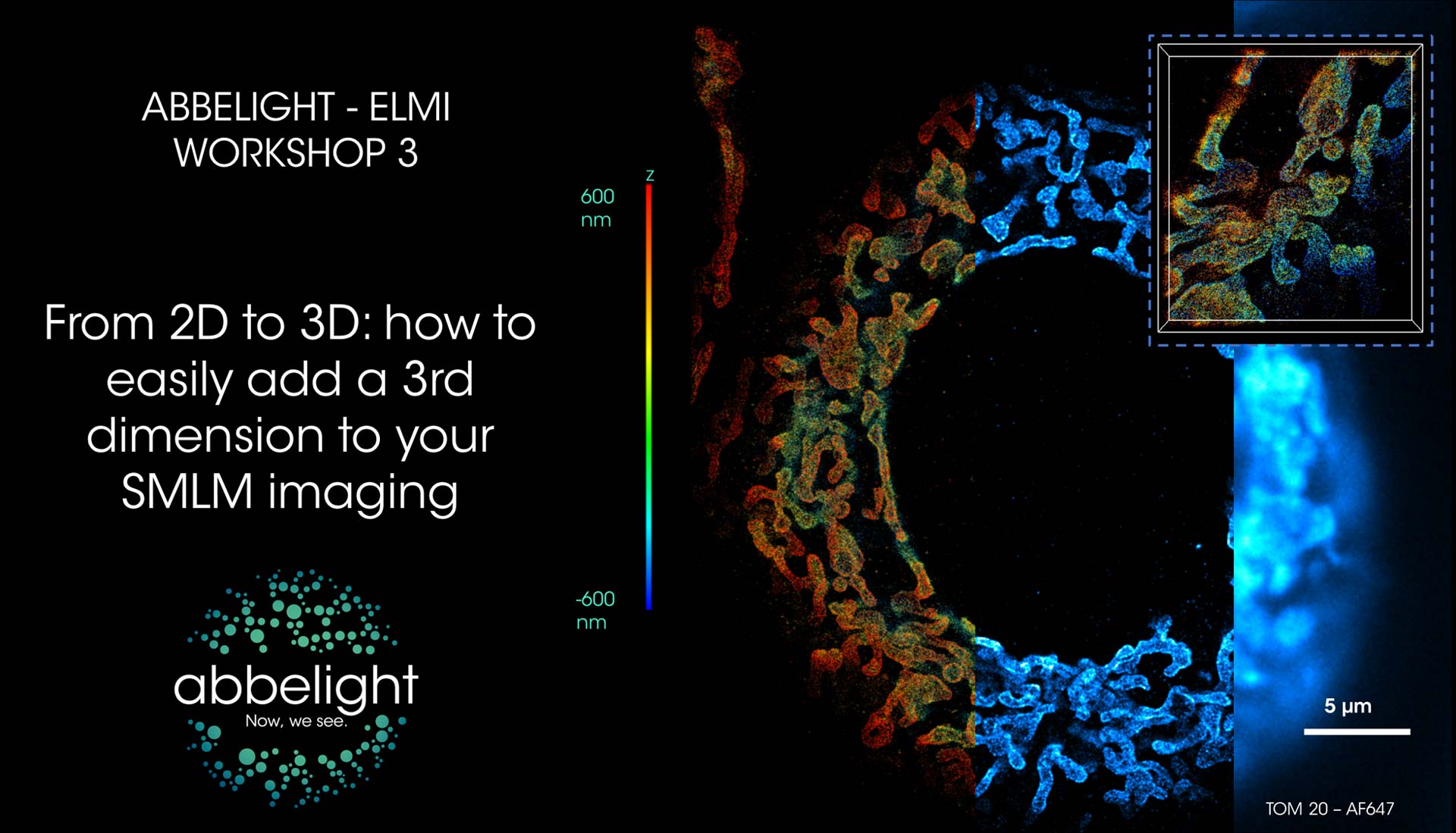
From 2D to 3D: how to easily add a 3rd dimension to your SMLM imaging
In the past years, single-molecule localization microscopy (SMLM) has produced stunning and ever more quantitative results; however, its widespread application is hampered by complexities of implementation and difficulties of commercial solutions in controlling sources of aberration, such as drift, chromatism and tilt.
Abbelight instruments are designed to put super-resolution microscopy within reach of non-specialists and fully realize the potential of SMLM methods. Of importance, is the ability to perform quantitative & reproducible 3D SMLM.
PSF engineering is a widely spread approach to encode the axial localization of molecules into the shape of their PSF. The simplest and most commonly used approach consists of using a cylindrical lens to apply an astigmatic aberration in the detection path. However, this comes at the cost of degrading the lateral localization precision of detected molecules as compared to those with unmodified PSFs.
The abbelight SAFe360 dual-camera nanoscopy module enables 3D imaging without compromising on the lateral resolution. Indeed, our technology takes advantage of 2 fluorescence paths that can be imaged simultaneously: one containing the z information encoded in the astigmatic PSF of individual molecules, and a second one containing an aberration-free image of the same molecule. This approach preserves the lateral resolution while simultaneously recording the axial information encoded in the deformed PSF.
In this workshop, we will demonstrate how to easily perform 3D calibration using beads, followed by recording of single color 3D SMLM images over a range of up to 1.2 microns using varied samples.
Schedule:
15 min Introduction: SMLM, astigmatism, SAFe360 (Presentation)
15 min Acquisition of calibration, explanation of the imaging parameters
5 min – Sample mounting
15 min – Acquisition of a 3D image and analysis
10 min – Summary of the workshop & Questions
ACQUIFER Imaging GmbH
Room: Gallery 1
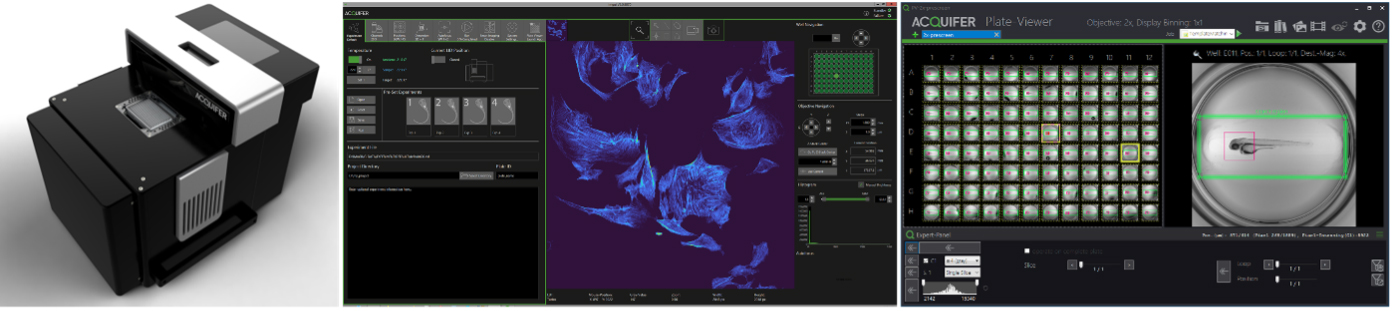
The ACQUIFER Imaging Machine – Workflows for Automated Imaging, Photomanipulation and High Content Screening
Phenotypic screening of complex cellular or whole organism model systems is increasingly employed in biomedical and pharmaceutical research. To fully exploit their potential, novel imaging workflows and tailored tools compatible with the automated imaging and spatiotemporal scoring of cell- and tissue-specific phenotypes are needed.
The ACQUIFER Imaging Machine (IM) is a versatile and flexible automated wide-field fluorescence microscope for high-content imaging of 2D/3D cell-cultures, fixed tissues sections and small model-organisms. The system features a unique moving optical unit in combination with a static sample holder, rendering it ideal for non-adherent and motion-sensitive specimen. For optimal time-lapse in-vivo imaging, the sample chamber is equipped with a Peltier device for highly precise temperature control.
In this workshop, we will demonstrate the unique hardware design and exemplify automated imaging applications. We will illustrate our workflow concept for automated imaging, feedback microscopy and High-Content-Screening using the ACQUIFER IM and associated software tools. Furthermore, we will discuss the optional laser-module that allows scaling-up of challenging photomanipulation experiments, such as photodamaging or photoconversion.
The IM control software ensures simple and intuitive configuration of standard imaging protocols. Complex acquisition procedures can be customized thanks to built-in scripting support (C#-based) or via dedicated extensions for external applications (Fiji, Python etc). The powerful imaging capabilities of the IM are complemented by the ACQUIFER PlateViewer, an easy-to-use software for browsing and visualization of large high-content screening datasets, and an interface for supervised feedback microscopy. The PlateViewer includes a plugin mechanism, allowing interfacing with external software for custom image-analysis. The open interface enables remote-control of the system by external software or integration in a robotic setup. It also allows feedback microscopy workflows for pre-scan/re-scan applications, such as automated centring applications, using Fiji and the PlateViewer.
Refer to our website at acquifer.de
ARGOLIGHT
Stand 9
Obtain and manage quality control data on your microscopes
One of the core facilities’ duties is to provide end-users, usually researchers in life sciences, a fleet of microscopes at a level of performance compatible with their experiments. This is not an easy task because the performance of such devices tends to fluctuate or deteriorate over time for many reasons: misusing, aging, environment fluctuations, etc. This is especially true for high end imaging systems such as confocal or super resolution microscopes. Having access to a unique, reliable, and easy-to-use device to ensure microscopes’ performance would certainly make easier this tedious task.
To get quantitative and reproducible data, assessing the performances of fluorescence microscopes is a prerequisite before any imaging campaign. For example, system co-registration accuracy should be evaluated before any co-localization study; System field uniformity and intensity response before any study where intensity in the image matters; Spatial resolution before any study aiming at counting objects close to each other, etc.
Three years after our last presence at ELMI, we are happy to come back and present during this workshop the novelties we have developed in the meantime: improved hardware products, new analyses from 3D patterns, new analyses related to non-Argolight products, improvements in user interface and user experience, etc. Above all, the presentation aims to show how the quality control of fluorescence microscopes can be standardized with Argolight software solutions, and how the generated quality control data can be managed and centralized for later reporting.
Bruker
Room: Goto 32
Imaging across scales – from single particle to whole living and cleared organisms
In this workshop we will demonstrate Bruker super-resolution and light-sheet microscopy solutions (Luxendo SPIM family).
The SPIM demonstration will cover different sample types and potential applications ranging from life imaging of delicate live specimen such as organoids and embryos (mouse, zebrafish etc. ) to whole in-toto imaging of large cleared samples such es entire adult mice. We will be showing our 3D Super-resolution system, the VXL with patented 3D technology suitable for STORM and PALM, along with an integrated
microfluidics system for highly multiplexed, DNA-PAINT research.
Workshop 1/4 : TruLive3D SPIM – light-sheet microscopy for live samples
Workshop 2/5 : VXL – 3D super-resolution microscopy
Workshop 3/6 : LCS SPIM – light-sheet microscopy for cleared samples
Image:
Top, left image: live zebrafish embryo H2A::GFP recorded on Luxendo MuVi SPIM LS. Top, right image: Mitochondria stained for TOM20 with Alexa 647 and imaged with STORM. Bottom, left image: cleared adult mouse with GFP labeled nerves and recorded on Luxendo LCS SPIM. Bottom, right: Cleared YFP expressing transgenic mouse brain recorded on Luxendo MuVi SPIM CS
Carl Zeiss Microscopy
Room: Goto 33
Discover the evolving technology of Lattice Lightsheet Microscopy
The launch of the ZEISS Lattice Lightsheet 7 inverted platform in 2020 was an important milestone for lattice lightsheet microscopy. This platform was designed for use by the non-expert so no alignment or technical know-how is needed to capture data. This has led to wide adoption of the Lattice Lightsheet 7 and it has now emerged as one of the key solutions for long term live imaging in the core facility. Further expansion of the Lattice Lightsheet 7 platform to more capability both in acquisition and analysis to assist in scaling experiments to higher throughput now further extend the imaging possibilities.
This workshop will focus on lattice lightsheet technology and will introduce the new capabilities that the ZEISS Lattice Lightsheet 7 offers. You will see the full workflow in action from capturing the data through to segmentation and quantitative analysis of the resulting datasets using arivis Vision 4D.
CrestOptics SpA
Stand 34
Two-fold enhanced spatial resolution in high and low mag objectives with DeepSIM
The DeepSIM is the first super-resolution (SR) microscope module that is compatible with any existing upright or inverted microscope with a camera port and is as easy to use as a confocal microscope enabling scientists to access SR data about their biological samples. Why DeepSIM? Resolution is doubled in all dimensions compared to a confocal microscope, reaching 100nm in XY and ~ 300nm in Z. In this workshop, we demonstrate that two-fold enhanced spatial resolution can be obtained with high and low magnification objective from 100X to 20X, making CrestOptics DeepSIM SR module a reliable, simple to use and affordable solution to study sub-cellular details and expand the range of applications.
#super-resolution #SIM
Cytena
Stand 8
Fluorescence in QPI Imaging with Livecyte: The mCherry on Top
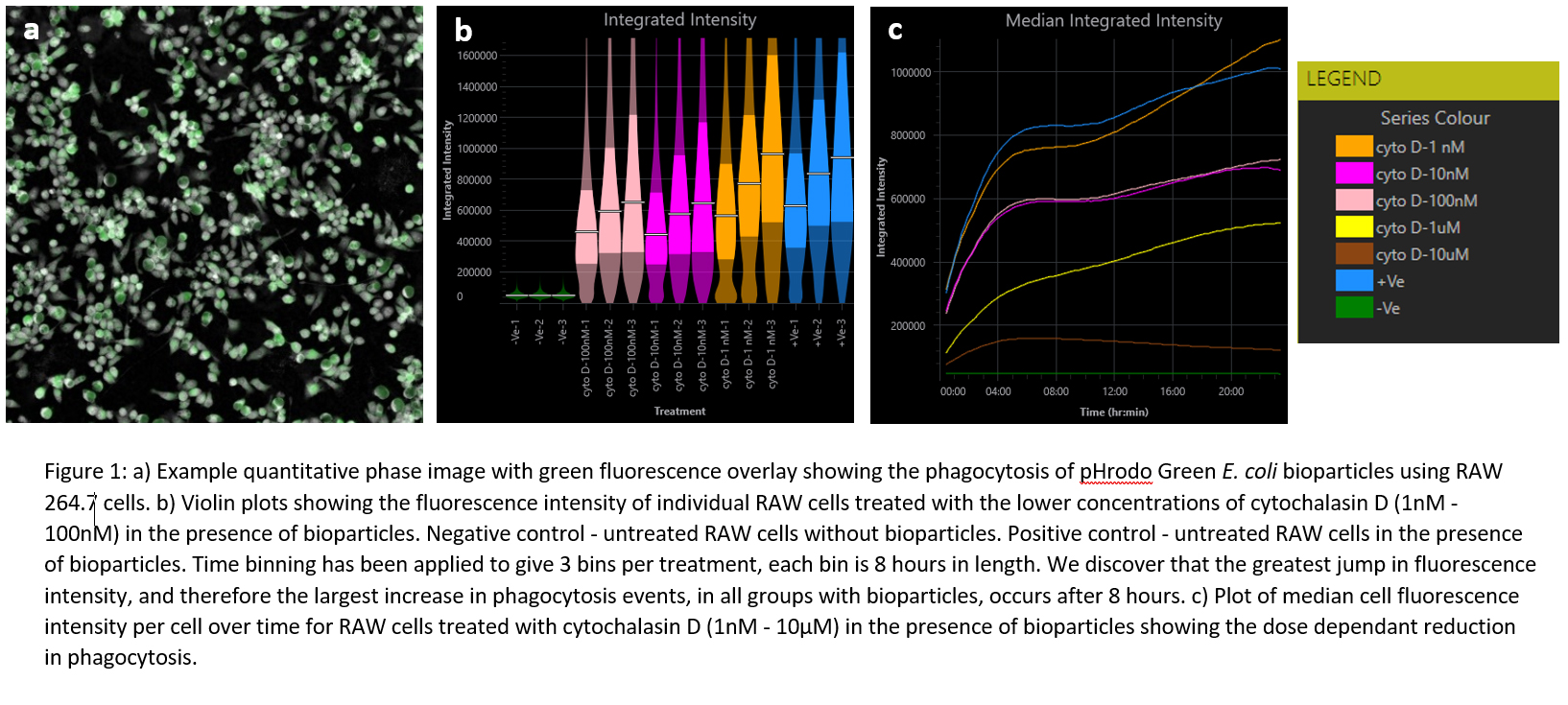
Fluorescent imaging can be a double-edged sword; beneficial in biological models, characterising subcellular detail and monitoring co-cultures [1], but at the same time playing havoc with the health of your cells. Livecyte’s correlative fluorescence capability enables fluorescence signal to be captured infrequently, but the signal from individual cells to be linked over time using the tracking data from frequent quantitative phase imaging; long-term fluorescence intensity whilst substantially reducing phototoxicity effects.
This workshop will guide users through Livecyte’s Analyse Fluorescence Dashboard and explore some of the unique metrics it can achieve. In particular, how we can investigate phagocytic activity in macrophages using fluorescent bioparticles in response to a cytotoxic drug cytochalasin D. The Fluorescence Dashboard in Livecyte’s Analyse software reveals single-cell metrics marking a dose dependant reduction in median fluorescent intensity of macrophages and therefore a reduction in phagocytosis.
Fluorescence is also useful in understanding subsets of cells within the same population. In this workshop we will investigate differences between differentiated and progenitor cells both in isolation and co-culture. Using the Explore page in Livecyte’s Analyse software, subpopulations of cells can be defined with gates or filters based on cell behaviour, morphology or fluorescence expression. Differentiated cells were found to be larger and have reduced motility compared to progenitor cells. It was also possible to determine changes in behaviour when cells were in co-culture compared to cells in a homogeneous population with differentiated cells proliferating at a slower rate when they are in isolation.
ibidi GmbH
Stand 29
ibidi Imaging Chambers and Surfaces
High-resolution microscopy of cells requires imaging chambers that are specifically tailored to your application.
Are you interested in learning about…
• … easy to handle all-in-one solutions for live cell imaging?
• … the advantages of using channel slides for immunofluorescence stainings?
• … solutions for 3D cell culture and spheroid imaging?
Join our workshop and find the answers to these questions!
First, we will introduce a variety of ibidi chambers with different geometries and surfaces and help you to choose the products that are best suited for your assay. This will be followed by a hands-on-training where you get the chance to test a selection of our labware for 2D and 3D cell culture and imaging.
Leica Microsystems CMS GmbH
Room: Teatro
STELLARIS: From Power HyD enhanced photon detection to molecular interactions with TauSense
Confocal microscopy has become a staple for fluorescence imaging in the life sciences, owing to the optical sectioning, sensitivity, and the temporal and spatial resolution capabilities. With STELLARIS, we have introduced key innovations such as the Power HyD family of detectors (1), the next generation of white light lasers (WLL), and the TauSense technology (2).
The fully digital and photon counting proprietary approach, Power Counting, allows straightforward and robust quantification of fluorescence signals in STELLARIS. Together with the WLL excitation flexibility, STELLARIS delivers extended near infrared capabilities for expanding the palette of fluorophores in every experiment., In addition, . TauSense opens the door to functional imaging insights thanks to straightforward access to fluorescence lifetime-based information.
In this workshop we will learn how all the technological innovation in STELLARIS facilitate fluorescence quantifications . We will also unveil a new TauSense tool built to evaluate molecular interactions. Join us for a first-hand view of what STELLARIS can bring to functional imaging.
1. V. Schweikhard, L. A. J. Alvarez, I. Steinmetz, M. J. Roberti, Holger Birk, A. Giske. The Power HyD family of detectors. Nat. Methods. 2020. Doi: d42473-020-00398-0 (2020).
2. M. J. Roberti, L. Ortiz Lopez, G. Ossato, I. Steinmetz, P. Haas, F. Hecht, and L. A. J. Alvarez. TauSense: a fluorescence lifetime-based tool set for everyday imaging. Nat Methods. 2020. Doi: d42473-020-00364-w
Miltenyi Biotec B.V. & Co. KG
Room: Gallery 2
Dive deep into your sample – the Miltenyi 3D imaging workflow
Visualizing the three-dimensional architecture of complex organisms while analyzing biological processes in a whole-organ fashion are becoming the new standard in research. To provide a complete, smooth, and hassle-free 3D imaging workflow, Miltenyi Biotec covers this entire process offering solutions for sample staining, clearing and imaging. In this workshop, we will guide you through the most important steps of sample preparation, including a live demonstration, and will demonstrate how easily high quality 3D images of large samples can be acquired with our cutting-edge light sheet system, the UltraMicroscope BlazeTM.
Nikon Europe BV
Room: Move 1
Tilt - A new angle on live cell imaging
The Tilt live cell imaging system is an add on to most any inverted microscope that significantly reduces photobleaching and phototoxicity in live cell imaging allowing for the observation of biologically relevant temporal dynamics. Working with both lower and high NA objectives, including 100x 1.49NA objectives, the Tilt is well suited for both organismal- based studies as well as the study of high resolution, intracellular dynamics.
Prospective Instruments
Room: Goto 31
First turnkey Multiphoton Microscope!
Bring your own sample to the conference to image it with Prospective Instruments MPX series Two-Photon Microscope. Contact us if you would like to take this opportunity.
Of course, we will also bring a selection of samples to image.
TissueGnostics GmbH
Room: Logi 1
Practical Application of Machine Learning in Tissue Cytometry - High-Plex Analyses
In September 2021 the United States’ Food and Drug Administration (US-FDA) has approved the first AI-based decision support system for prostate cancer diagnostics. This hallmark indicates a historic decision as it is the first time in the history of medicine that a regulatory body has accepted a software-only solution, which analyses microscopic images by using artificial intelligence!
This indicates both, that technologies reach a performance and maturity level that makes diagnostic routine applications not only possible but also feasible and that the market demand for such solutions has reached a level where it has become viable for industry to invest in the development of commercial solutions as a return on investment can be expected.
The TissueFAXS Cytometry platform incorporates Machine & Deep Learning algorithms. It can do end-point assays as well as live-cell imaging and time-kinetic experiments. TissueFAXS Cytometry also promotes tissue cytometry to a new level of quality, where complex cellular interactions can be addressed on the single-cell level but still in histological context.
In this workshop we will give practical examples of high-end tissue cytometry applications using Deep & Machine Learning in TissueGnostics’ flagship application StrataQuest.
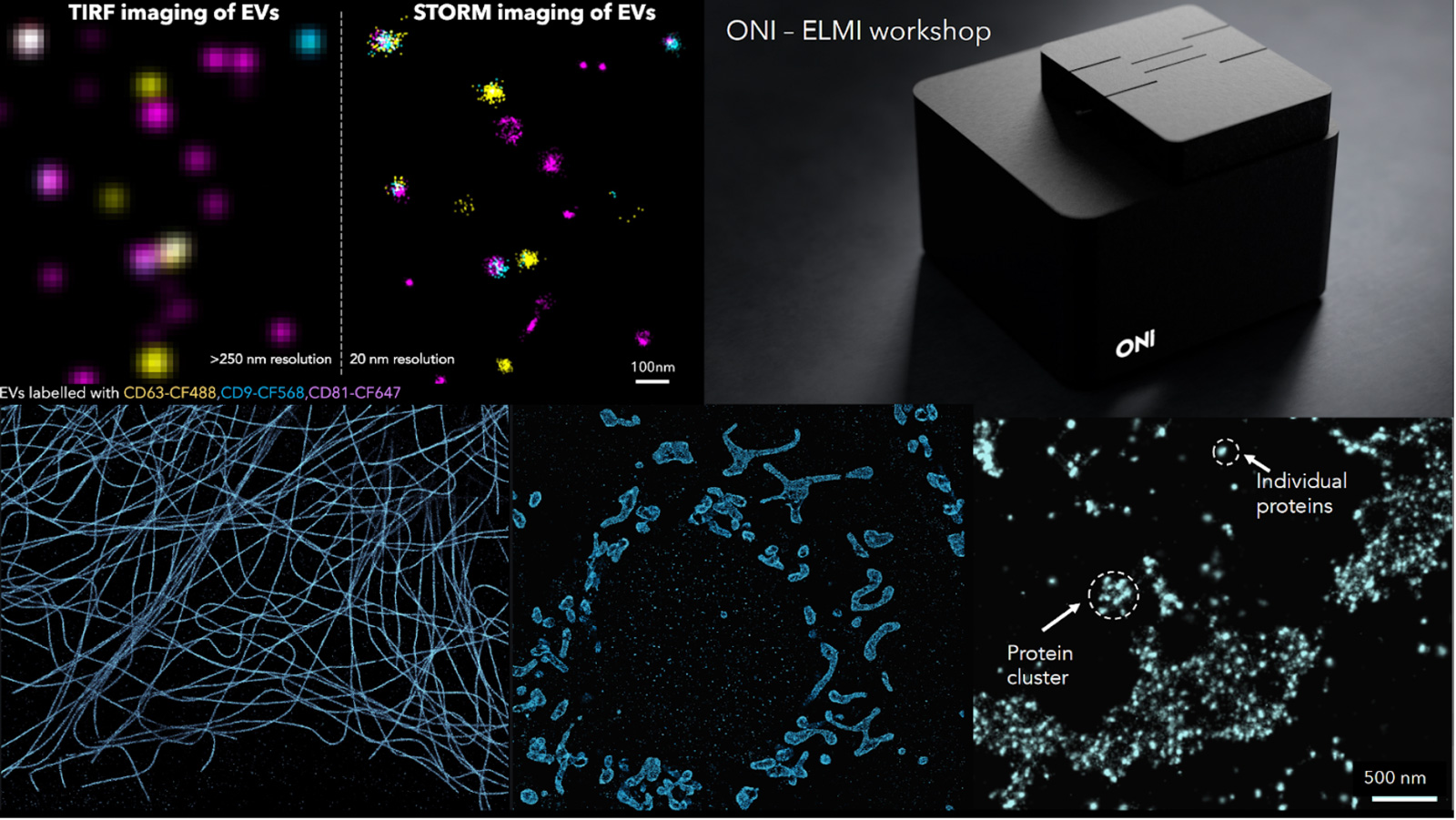
ONI
Room: Move 2
Every Molecule Counts - the ONI super-resolution platform just got even better
Studying spatial protein organization or protein copy number, clustering and mobility at the molecular level is extremely important from both a diagnostic and treatment perspective. Looking at cellular structures beyond the diffraction limit with a resolution of 15-20 nm with robust quantification has been enabled with Single-molecule localization microscopy (SMLM) like STORM or PALM. ONI has created the world’s first desktop super-resolution microscope, the Nanoimager, which is designed to operate on a standard lab bench and has a footprint smaller than a piece of A4 paper, with the most intuitive and simple software interface, making it also accessible to researchers not specialized in high end microscopy techniques with one major goal: make it accessible and simple to use.
It is really important to enable users to extract quantitative information from their localization-based images. We have done this by designing a cloud-based analysis software called CODI (COllaborative DIscovery). CODI helps users analyze their single-molecule data by extracting quantitative information on protein clustering, cluster size and area, cluster density, and so much more. It enables all researchers to upload their data, share or collaborate with other scientists, whilst gaining meaningful insights about their data.
During this workshop we will perform a two-color STORM on mitochondrial samples (TOM20 and HSP60) and reveal the sub-mitochondrial compartmental organization with 20 nm resolution. The aim is to introduce SMLM imaging as a powerful tool for rapid and multiplexed characterization of cellular structures along with a visual and quantification analysis platform CODI developed by us. We will follow up the workshop with an open discussion on the wide-ranging applicability of the Nanoimager in different biological applications encompassing the field of immunology, neurobiology, cell biology and cancer.
Visitron Systems GmbH
Stand 27
VISITIRF ORBITAL-RINGTIRF TECHNOLOGY
VISIVIEW 6.0
VISIVIEW IMAGING SOFTWARE WITH NEW REALTIME 5D IMAGE ACQUISITION AND DISPLAY VIRTEX REALTIME EXPERIMENT CONTROL
This workshop will give the listener an overview of new developments in RingTIRF technology, new features in VisiView Imaging Software and highly accurate experiment control by ViRTEx.
2. ORBITAL Ring-TIRF Technology
The VisiTIRF-ORBITAL is a compact and powerful high speed 2D galvo driven spinning Ring-TIRF laser illumination system. It offers a large and evenly illuminated field of view to enable applications such as single molecule tracking or SMLM – Single Molecule Localisation Microscopy for superresolution imaging. Full 360 degree positioning by free circular diameter or elliptical trajectory at the back focal plane of the high aperture TIRF objective offers illumination with minimal fringes or shading gradients